The year in cardiovascular medicine 2021: interventional cardiology
█ Current opinion
Authors:
Javier Escaned1, Farouc A. Jaffer2, Julinda Mehilli3, Roxana Mehran4
1Hospital Clínico San Carlos IDISCC, Complutense University of Madrid, Madrid, Spain
2Cardiology Division, Department of Medicine, Massachusetts General Hospital and Harvard Medical School, Boston, MA, USA
3Medizinische Klinik I, Landshut-Achdorf Hospital, Landshut, Klinikum der Universitaet München,
Ludwig-Maximilians-Universitaet and German Centre for Cardiovascular Research (DZHK), Partner Site Munich Heart Alliance, Munich, Germany
4The Zena and Michael A. Wiener Cardiovascular Institute, Icahn School of Medicine at Mount Sinai, New York, NY, USA
Corresponding author. Tel: +34 91 3303438, Fax: +34 91 3303289, Email: escaned@secardiologia.es
Preamble
Since last year’s report in the European Heart Journal, we have witnessed substantial progress in all aspects of interventional cardiology. Of note, the practice of interventional cardiology took place amidst successive waves of the COVID-19 pandemic, which continues to be a major burden for all healthcare professionals around the globe. In our yearly review, we shall revisit the developments in percutaneous coronary intervention (PCI), structural heart interventions, and adjunctive pharmacotherapy.
ISSUE: CARDIOLOGIA HUNGARICA | 2023 | VOLUME 53, ISSUE 1
Introduction
Since last year’s report in the European Heart Journal, we have witnessed substantial progress in all aspects of interventional cardiology. Of note, the practice of interventional cardiology took place amidst successive waves of the COVID-19 pandemic, which continues to be a major burden for all healthcare professionals around the globe. In our yearly review, we shall revisit the developments in percutaneous coronary intervention (PCI), structural heart interventions, and adjunctive pharmacotherapy (Graphical Abstract).
Non-culprit lesion revascularization in ST-segment elevation myocardial infarction
Revascularization in multivessel coronary disease
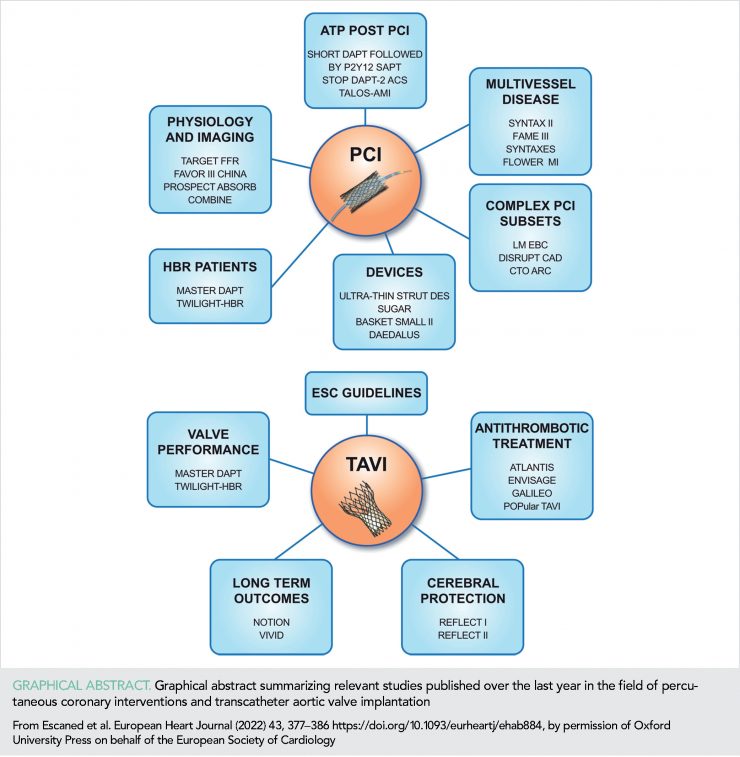
The 5-year outcomes of the SYNTAX II trial showcase the benefits of integrating best practices of PCI (imaging- and physiological-PCI guidance, thin-strut stents, and more complete revascularization) into a single revascularization strategy (SYNTAX II strategy) in treating patients with three-vessel coronary disease (3VD) (7). At 5 years, major adverse cardiac and cerebral events (MACCEs) in SYNTAX II were significantly lower than in a matched cohort of SYNTAX-I PCI patients (21.5 vs. 36.4%, P <0.001) (Figure 1), with lower rates of revascularization (13.8 vs. 23.8%, P <0.001), and MI (2.7 vs. 10.4%, P <0.001), consisting of both procedural MI (0.2 vs. 3.8%, P <0.001) and spontaneous MI (2.3 vs. 6.9%, P = 0.004).
All-cause mortality was lower in SYNTAX II (8.1 vs. 13.8%, P = 0.013) reflecting a lower rate of cardiac death (2.8 vs. 8.4%, P <0.001). Major adverse cardiac and cerebral event outcomes at 5 years among patients in SYNTAX II and pre-defined patients in the SYNTAX-I coronary artery bypass graft (CABG) cohort were similar (21.5 vs. 24.6%, P = 0.35).
The results of the FAME III randomized clinical trial (RCT), which compared from a non-inferiority standpoint of the clinical outcomes of 1500 patients with 3VD randomized to either the FFR-guided PCI or CABG, were published (8). At 1-year follow-up, MACCE rate in FAME III was 10.6 and 6.9% among patients assigned to PCI and CABG, respectively [hazard ratio (HR) = 1.5, 95% confidence interval (CI: 1.1–2.2)]; thus, non-inferiority of the FFR-guided PCI was not reached (P = 0.35 for non-inferiority) (Figure 1). Notwithstanding the differences in the study design, at first glance, the results of FAME III seem discordant with those of SYNTAX II. However, it is noteworthy that FAME III explored the specific value of the FFR-based revascularization, and not that of an array of clinical practices (including imaging- and physiological-guidance) encompassed into the SYNTAX II strategy.
Thus, at a difference with SYNTAX II, FAME III enrolled patients with CABG recommended as the preferred treatment according to the SYNTAX score II, and intracoronary imaging was seldom used (12 vs. 87% in SYNTAX II). Future randomized studies of CABG vs. PCI might focus on whether integrated best practices (i.e. heart team-based patient stratification, use of intracoronary physiology and imaging, PCI competence in anatomical complex subsets, and optimal medical treatment) in patients with MVD.
Based on the SYNTAX Extended Survival study (SYNTAXES), a new index, named SYNTAX score II 2020 (SS II 2020) was derived to perform the prediction of 10-year outcomes after PCI or CABG in patients with MVD. The SS II 2020 demonstrated a discriminative ability in the PCI and CABG groups for predicting 10-year all-cause deaths [C-index = 0.73, (95% CI: 0.69–0.76) for PCI and C-index = 0.73, (95% CI: 0.69–0.76) for CABG] and 5-year major adverse cardiovascular events (MACEs) [C-index = 0.65, (95% CI: 0.61–0.69) for PCI and C-index = 0.71, (95% CI: 0.67–0.75) for CABG]. The index has the potential of supporting heart teams, patients, and their families in selecting the optimal revascularization modality (9). The SYNTAXES study also reported that female sex was not an independent predictor of mortality at 10 years in patients with MVD (10) and stressed the importance of optimal medical treatment after coronary revascularization (11).
New drug-eluting stents and drug-coated balloons
The impact of technological improvements in drug-eluting stents (DES) has been the focus of several publications. Whether the reduction in strut thickness improves PCI outcomes was investigated in a study-level meta-analysis of 16 RCTs (20 701 patients) comparing an ultrathin-strut DES to a conventional second-generation thin-strut DES (12). At a mean follow-up of 2.5 years, the ultrathin-strut DES use reduced the risk of TLF, driven by less cardiac death-target lesion revascularization (CD-TLR) compared with the conventional second-generation thin-strut DES, with similar risks of MI, ST, cardiac death, and all-cause mortality.
The SUGAR RCT investigated the value of the Cre8 EVO stent, designed to release sirolimus with an amphiphilic carrier from laser-dug wells, in diabetic patients undergoing PCI. A total of 1175 patients were randomized to PCI with either Cre8 EVO or zotarolimus-eluting Resolute Onyx stents. At 1-year follow-up, the study revealed that Cre8 EVO stents were non-inferior to Resolute Onyx stents in terms of target lesion failure [7.2% in Cre8 EVO and 10.9% in Resolute Onyx arms, HR = 0.65, (95% CI: 0.44–0.96); P non-inferiority <0.001]. An exploratory analysis for superiority at 1 year suggested the superiority of the Cre8 EVO over Resolute Onyx stents (13).
Treatment of small coronary vessels remains a major challenge for PCI, and the use of stent-avoidance strategies has been considered in this context. A pre-specified substudy of the BASKET-SMALL-2 trial investigated 758 patients randomly assigned to drug-coated balloon (DCB) or DES treatment of de novo lesions in vessels <3 mm diameter. The Kaplan–Meier MACE rate estimate was 15% in both groups [HR = 0.99, (95% CI: 0.68–1.45); P = 0.95]. The study revealed the maintained efficacy and safety of DCB vs. DES in the treatment of de novo coronary small vessel disease up to 3 years (14). In the context of in-stent restenosis (ISR), the recent DAEDALUS meta-analysis examining DCB-percutaneous transluminal coronary angioplasty vs. DES to treat DES-ISR demonstrated a significantly higher 3-year repeat revascularization rate in the DCB group [HR = 1.58, (95% CI: 1.16–2.13)] (15).
Complex coronary lesion subsets
The EBC-LM RCT enrolled patients with left main (LM) stenosis involving its bifurcation to a stepwise provisional strategy (n = 230) or a systematic dual stent approach (n = 237) (16). The composite endpoint of death, MI, and TLR at 12 months occurred in 14.7% of the stepwise provisional group vs. 17.7% of the systematic dual stent group [HR = 0.79, (95% CI: 0.5–1.3); P = 0.34)] (Figure 2).
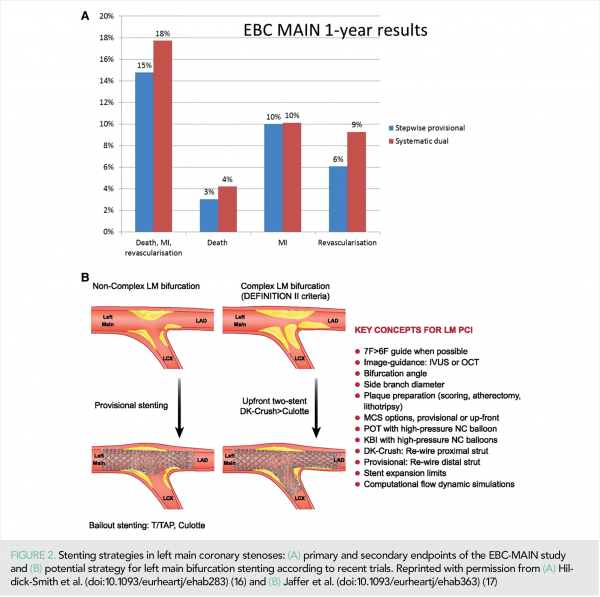
Procedure time, X-ray dose, and consumables favoured the stepwise provisional approach. Symptomatic improvement was excellent and equal in each group. These findings support using a stepwise provisional strategy as default for distal LM stem bifurcation PCI, although lower side branch complexity was present compared with the DK-CRUSH IV trial (16, 17).
Two relevant consensus documents in the field of chronic total occlusion (CTO) PCI have been published. The first, the CTO-ARC recommendations, intend to standardize key elements and procedural definitions, endpoint definitions, and clinical trial design principles in the field of revascularization of CTOs (18). The second is a proposal of a global CTO crossing algorithm aimed to facilitate decision-making and CTO PCI teaching across various geographies and improve the safety, reproducibility, and efficiency of these procedures (19).
Two registry studies provided contemporary insights into the use and outcomes of atherectomy for calcified lesions. First, an examination of the National Cardiovascular Data Registry (NCDR) CathPCI demonstrated that atherectomy rates tripled in the USA from 1.1% in 2009 to 3.0% in 2016, with a concomitant temporal decline in MACE rates [odds ratio (OR) = 0.98, (95% CI: 0.97–0.99)] but increase in coronary perforation rates [OR = 1.18, (95% CI: 1.04–1.35)] (20). In an analysis of 7740 rotational atherectomy procedures from the British Cardiovascular Intervention Society national PCI database, a significant inverse association was observed between atherectomy PCI volume and in-hospital mortality [OR = 0.986, (95% CI: 0.975–0.996)] as well as MACCE [OR = 0.983, (95% CI: 0.975–0.993)] (21).
Additional information on the safety and effectiveness of intravascular lithotripsy (IVL) was obtained in the DISRUPT CAD III study, a prospective, single-arm multicentre investigation designed for regulatory approval of coronary IVL, enrolling 431 patients with severely calcified de novo coronary lesions undergoing PCI. At 30-day follow-up, freedom from the MACE was 92.2%; the lower bound of the 95% CI was 89.9%, exceeding a pre-specified performance goal (PG) of 84.4% (P < 0.0001). The primary effectiveness endpoint of the procedural success was 92.4%; the lower bound of the 95% CI was 90.2%, which exceeded the PG of 83.4% (P < 0.0001) (22). Percutaneous coronary intervention guidance with intracoronary physiology and imaging As part of the renewed interest in performing wire-based functional assessment of PCI results, the TARGET FFR RCT investigated the feasibility and efficacy of an FFR-guided optimization strategy in achieving post-PCI FFR values of >0.90, compared with the standard optimization based on angiography (23). The study, which included 260 patients, demonstrated a high rate (68.1%) of post-PCI FFR values >0.90. In the FFR-guided arm, further intervention in 30.5% patients did not significantly increase the proportion of patients with a final FFR >0.90, but reduced the proportion of patients with a final FFR < −0.80, compared with the angiography-guided arm [−11.2%, (95% CI: −21.87 to −0.35); P = 0.045]. The 5-year outcomes of the IVUS-XPL RCT, which enrolled patients with lesions ≥28 mm undergoing DES PCI, demonstrated a sustained reduction in MACE rates in the IVUS-guided group compared to the angiography-alone group [HR = 0.50, (95% CI: 0.34–0.75)].
Benefits of IVUS were driven by reductions in TLR rates (24). The results of the first RCT investigating the clinical benefit of using functional coronary angiography (wireless, angiography-derived flow reserve) to guide PCI, FAVOR III China, has generated great interest (25). This sham-controlled RCT enrolled 3825 patients with chronic coronary syndrome or ACS to compare the clinical outcomes (MACE) of a quantitative flow ratio (QFR) strategy (PCI performed whenever the QFR is ≤0.80) or an angiography-guided strategy (PCI performed based on the visual angiographic assessment). At 1-year follow-up, MACE rate was 5.8 and 8.8% in the QFR- and angio-guided arms, respectively [HR = 0.65, (95% CI: 0.51–0.83); P = 0.0004]. High-risk vulnerable plaque Several studies provided new evidence on the diagnosis and interventional management of vulnerable plaques. In an analysis of 1497 trials, CAD patients undergoing serial IVUS over 18–24 months, progression of IVUS-detected attenuation or echolucency associated with a higher rate of cardiovascular events, supporting a potential role IVUS identification of high-risk vulnerable plaques (26).
The IVUS imaging further provided the foundation for the preventative PCI PROSPECT-ABSORB trial (27). Patients (n = 185) with lesions with >65% plaque burden were randomized to medical therapy or PCI with the biodegradable drug-eluting ABSORB scaffold. Target lesion failure rates (primary endpoint) were similar in both groups of ~4.4% at 24 months (P = 0.96), although the study was not powered for clinical endpoints. The secondary endpoint of the lesion-related MACE trended in favour of the PCI group (P = 0.12). Thus, the study provides favourable evidence to support future RCTs on this topic powered to draw conclusive results
The debate on whether lesion biology, and not its ischaemia-generating character, impacts prognosis has been warmed-up by the results of the COMBINE OCT-FFR study, a prospective, double-blind investigation focused on the management of ACS non-culprit stenosis in diabetic patients. Whenever the FFR was >0.80, PCI was deferred and an optical coherence tomography was performed to assess the presence or absence of thin-cap fibroatheromas (TCFA). Subsequently, the outcomes of TCFA-positive patients were compared with those of TCFA-negative patients (28). Patients with FFR-negative TCFA-positive plaques comprised 25% of the cohort, and exhibited significantly higher 18-month MACE rates than those with FFR-negative, TCFA-negative plaques [HR = 4.65, (95% CI: 1.99–10.8)], consistent with the earlier CLIMA study. Overall, it is foreseeable that larger studies of preventative PCI of high-risk lesions identified by intravascular imaging will be launched based on these studies.
Transcatheter treatment of aortic valve stenosis
The catheter-based aortic valve procedure has long been entered in the treatment armamentarium of aortic valve stenosis. Publications of the last year in this field are focused on RCTs comparing different transcatheter valvular bioprostheses, on testing different approaches to increase the safety of transcatheter aortic valve implantation (TAVI) procedures, long-term outcomes after TAVI or surgical aortic valve replacement (SAVR), as well as the update of 2021 ESC/EACTS Guidelines for the management of valvular heart disease.
Performance of different bioprosthetic valves
The newest generation of balloon-expandable Sapien valve (BEV) and self-expandable CoreValve Evolute valve (SEV) has been randomly compared within the setting of the SOLVE-TAVI (compariSon of secOnd-generation self-expandable vs. balloon-expandable Valves and gEneral vs. local anesthesia in Transcatheter aortic Valve Implantation) trial. These two modern valve systems perform similar regarding the combined endpoint of all-cause mortality, stroke, moderate or severe paravalvular leakage, and permanent pacemaker implantation at 1 year [HR = 0.94, (95% CI: 0.70–1.26); P = 0.66] (29).
In the SCOPE-2 RCT (Safety and Efficacy Comparison of Two TAVI Systems in a Prospective Randomized evaluation-2), the recent generation of SEV was compared with the first-generation ACURATE neo bioprosthesis. In this study of 796 patients, the non-inferiority of ACURATE neo to SEV regarding the 1-year incidence of all-cause death or stroke was not proven (15.8 vs. 13.9%, absolute risk difference 1.8%, upper one-side 95% confidence limit 6.1%; P = 0.0549 for non-inferiority). The higher rate of residual moderate or severe aortic regurgitation with the ACURATE neo compared with the SEV (10.0 vs. 3.0%, P = 0.002) might have contributed to this result (30). With the newest generation, ACURATE neo 2 bioprosthesis, the rate of moderate or severe aortic regurgitation is clearly reduced up to 2.5% at 1-year follow-up (31).
Technical features to enhance
transcatheter aortic valve implantation procedure safety
Increasing operators’ experience and advances in TAVI devices led to a decline in peri-TAVI stroke rates, thus, improving the TAVI procedure safety. On the other hand, silent ischaemic brain injury was detected by diffusion-weighted magnetic resonance effects in >80% of the TAVI patients (32). Aiming to avoid clinical and silent brain injury, two important RCTs have been published evaluating the performance of cerebral-protection devices during TAVI procedures. In the REFLECT I (Reduce the Impact of Cerebral Embolic LEsions after TransCatheter Aortic Valve ImplanTation) trial, TriGuard™ HDH cerebral-protection device was randomly used during TAVI. Only 68.8% (n = 258) of the planned patients were enrolled. The primary efficacy endpoint was a hierarchical composite of all-cause mortality or any stroke at 30 days, National Institutes of Health Stroke Scale worsening at 2–5 days or Montreal Cognitive Assessment worsening at 30 days and total volume of cerebral ischaemic lesions detected by diffusion-weighted magnetic resonance imaging at 2–5 days. Neither the primary hierarchical efficacy endpoint (mean efficacy score, higher is better: −5.3±99.8 TriGuard vs. 11.8±96.4 control, P = 0.31) nor the incidence of silent central nervous system injury was significantly different between both treatment strategies (32).
These results were supported by the next REFLECT II trial, which enrolled 220 patients (63.4% of the planned patients) and randomly compared in 2:1 fashion use or not use of the TriGuard 3 embolic protection device during TAVI. Again, the pre-specified primary superiority efficacy endpoint was not met [mean scores (higher is better): −8.58 TG3 vs. 8.08 control; P = 0.857) (33). Findings of these studies suggest a selective use of cerebral-protection devices during TAVI.
Balloon dilatation [balloon aortic valvuloplasty (BAV)] prior to valve implantation might increase the risk of annulus rupture, embolization, and haemodynamic instability. Thus, avoiding it might be attractive to simplify the TAVI procedure and probably increase the procedure safety. In the DIRECTAVI [TAVI Without Balloon Predilatation (of the Aortic Valve) SAPIEN 3] trial, direct TAVI without BAV was randomly compared with the conventional strategy using BAV with new-generation BEV (34).
The rate of device success in direct TAVI was non-inferior to that of BAV before the TAVI group [80.2 vs. 75.7%, mean difference 4.5%, (95% CI: 4.4–13.4); P = 0.02 for non-inferiority]. Few patients needed an unplanned BAV before TAVI, suggesting an anatomy-related upstream selection of patients in need of BAV before TAVI (34).
Long-term outcomes after transcatheter aortic valve implantation or surgical aortic valve replacement
Important insights about the long-term comparative performance of TAVI and SAVR among low-risk patients with aortic valve stenosis were derived from the NOTION RCT. In a TAVI population with a mean age of 79.1±4.8 years and a mean STS score of 3.0±1.7%, the 8-year estimated risks for all-cause mortality (51.8 vs. 52.6%; P = 0.90), stroke (8.3 vs. 9.1%; P = 0.90), or MI (6.2 vs. 3.8%; P = 0.33) were similar after TAVI and SAVR (Figure 3).
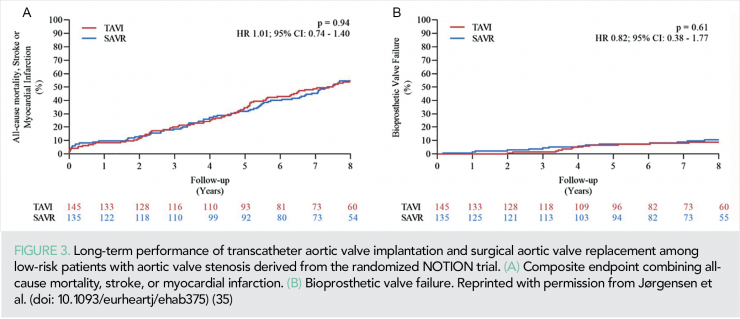
Whereas, the risk of structural valve deterioration was lower after TAVI than after SAVR (13.9 vs. 28.3%; P = 0.0017) (35). As TAVI now is indicated for low-risk patients with longer life expectancy, these long-term results are reassuring for TAVI in terms of clinical outcomes and valve durability.
Another continuously growing TAVI population is the collective of patients undergoing TAVI for failed surgical bioprosthesis. For the first time, the investigators of the international VIVID registry reported an 8-year follow-up data of patients undergoing Valve-in-Valve (ViV) procedures. Survival was lower among patients with small-failed bioprostheses compared with those with large-failed bioprostheses (33.2 vs. 40.5%, P = 0.01). In addition, independent predictors of reinterventions after ViV procedures were pre-existing severe prosthesis–patient mismatch, device malposition, use of balloon-expandable valves, and patients’ age. Thus, operator decision during both original SAVR and the ViV procedure might substantially influence clinical outcomes (36).
Regarding the treatment of aortic valve stenosis, the new ESC/EACTS Guidelines (37) highlight the importance of interaction heart team and patient regarding the aortic valve repair strategy selection (Class I, LOE C). Furthermore, alongside the STS risk score, patient’s age alone is now recommended as a determinant factor for TAVI or SAVR selection with TAVI recommended in patients aged ≥75 years (independent of operative risk score; Class I, LOE A). Another novelty is the recommendation of aortic valve repair in asymptomatic patients with severe aortic stenosis and left ventricular dysfunction (left ventricular ejection fraction <50%) without another cause (Class I, LOE B).
Adjunctive pharmacotherapy in interventional cardiology
Landmark studies have been published this year in the field of antiplatelet therapy after PCI. The MASTER DAPT study (38) was the first open-label RCT evaluating an abbreviated dual antiplatelet therapy (DAPT) duration vs. a standard regimen among patients with high bleeding risk (HBR) undergoing PCI. Between 30 and 45 days after PCI, 4579 event-free subjects were randomly assigned to single antiplatelet therapy (SAPT) with either P2Y12 or aspirin, or to a DAPT for at least 5 additional months (6 months after the index procedure). At 12 months after PCI, the short DAPT strategy was not inferior to the standard DAPT in terms of MACCEs, and was associated with a significant BARC (Bleeding Academic Research Consortium) 2, 3, or 5 bleeding risk reduction [−2.82%, (95% CI: −4.40 to –1.24); P < 0.001].
In the STOPDAPT-2 ACS trial (39), patients undergoing PCI for ACS, 1-month DAPT followed by clopidogrel monotherapy for 11 months did not meet the criteria for non-inferiority compared with a 12-month DAPT for the composite of cardiovascular death, MI, stroke, stent thrombosis, thrombolysis in myocardial infarction major or minor bleeding [3.2 vs. 2.8%; HR = 1.14, (95% CI: 0.80–1.62); P = 0.06]. Secondary outcome analyses revealed a higher incidence of nearly all ischaemic events within the short DAPT group, with only marginal gain in terms of bleeding risk. However, a meta-analysis (40) including 32 145 patients from five RCTs confirmed that a DAPT of ≤3 months followed by P2Y12 monotherapy was associated with a 37% relative risk reduction of bleeding [HR = 0.63, (95% CI: 0.45–0.86); P = 0.004] and a similar rate of fatal and ischaemic events in patients undergoing PCI with a second-generation DES. Additionally, in two prospective studies of HBR patients undergoing PCI (mainly for chronic coronary syndrome), a short DAPT of 1 or 3 months followed by aspirin monotherapy was non-inferior in terms of death or MI and was associated with a lower incidence of major BARC 3 or 5 bleeding when compared with a historical cohort receiving up to 12-month DAPT after propensity score stratification (41, 42). In the TWILIGHT-HBR substudy, ticagrelor monotherapy after a 3-month DAPT resulted in similar ischaemic outcomes and a large absolute reduction in major BARC 3 or 5 bleeding compared with a prolonged DAPT, among HBR patients undergoing PCI, two-thirds of whom for a non-STE ACS (43). This new evidence altogether indicates that a short DAPT course is a safe and effective bleeding-avoidance strategy, especially among HBR patients, but extra caution is warranted among those presenting with ACS.
A uniform unguided DAPT de-escalation of ticagrelor to clopidogrel was investigated in patients with acute MI and event-free during the first month after PCI in the TALOS-AMI trial (44). De-escalation significantly decreased the risk of net adverse clinical events up to 12 months [4.6 vs. 8.2%, HR = 0.55, (95% CI: 0.40–0.76); P = 0.0001] mainly by reducing bleeding complications. In the HOST-EXAM trial (45), patients who had undergone PCI 6–18 months prior were randomly assigned to a 24-month maintenance therapy with clopidogrel or aspirin monotherapy. The risk of a composite of all-cause death, non-fatal MI, stroke, readmission due to ACS, or BARC bleeding type 3 or greater was significantly reduced with clopidogrel [5.7 vs. 7.7%, HR = 0.73, (95% CI: 0.59–0.90); P = 0.0035], despite a numerically higher rate of fatalities.
Concerning the antithrombotic treatment in patients undergoing TAVI, the ATLANTIS (46) failed to demonstrate the superiority of a full-dose apixaban when compared with the current standard of care in patients with or without oral anticoagulation (OAC) indication, who have undergone a successful TAVI, with respect to the primary endpoint of death, stroke, MI, systemic emboli, intracardiac or valve thrombosis, deep vein thrombosis/pulmonary embolism, or major bleeding [18.4 vs. 20.1%, HR = 0.92, (95% CI: 0.73–1.16)].
These results, together with the previous GALILEO (47) and POPular TAVI (48) trials, suggest that the antiplatelet monotherapy should remain the default antithrombotic agent when there is no other indication for DAPT or systemic anticoagulation. In the ENVISAGE study (49), TAVI patients, with an indication to OAC mainly for atrial fibrillation, experienced similar (i.e. non-inferior) net adverse clinical events (including death from any cause, MI, ischaemic stroke, systemic thromboembolism, valve thrombosis, or major bleeding) with edoxaban when compared with vitamin K antagonists [17.3 vs. 16.5 per 100 person/years, HR = 1.05, (95% CI: 0.85–1.31), P = 0.93], despite a higher incidence of major bleeding [9.7 vs. 7.0 per 100 person/years, HR = 1.40, (95% CI: 1.03–1.91); P = 0.93 for non-inferiority].
Final outlook
The evidence reported over the last year in the field of interventional cardiology discussed in this review will likely be reflected in upcoming clinical practice guidelines and trigger new studies. Ongoing studies in functional coronary angiography (50) will complement the disruptive evidence provided by the FAVOR III China trials. The growing interest in integrating plaque biology and ischaemic burden to predict outcomes of patients with coronary stenosis will surely trigger debates and further research. Major studies on transcatheter structural heart interventions other than the TAVI are expected in the near future (51).
Conflict of interest
J.E. reports consultancies and/or speaker at educational events by Abbott, Abiomed, Boston Scientific, ShockWave, and Philips. F.A.J. has conducted sponsored research from Canon, Siemens, Shockwave, Teleflex, Mercator, and Boston Scientific; he is a consultant for Boston Scientific, Siemens, Biotronik, Magenta Medical, Asahi Intecc, IMDS, and Philips; and has equity interest in Intravascular Imaging, Inc. and DurVena. Massachusetts General Hospital has a patent licensing arrangement with Terumo, Canon, and Spectrawave; and F.A.J. has the right to receive royalties. J.M. declares receiving lecture fees from Astra Zeneca, Edwards LifeScience, Boston Scientific, Medtronic, BMS, and institutional research grant from Boston Scientific. R.M. reports institutional research grants from Abbott, Abiomed, Applied Therapeutics, Arena, AstraZeneca, Bayer, Biosensors, Boston Scientific, Bristol-Myers Squibb, CardiaWave, CellAegis, CERC, Chiesi, Concept Medical, CSL Behring, DSI, Insel Gruppe AG, Medtronic, Novartis Pharmaceuticals, OrbusNeich, Philips, Transverse Medical, Zoll; personal fees from ACC, Boston Scientific, California Institute for Regenerative Medicine (CIRM), Cine-Med Research, Janssen, WebMD, SCAI; consulting fees paid to the institution from Abbott, Abiomed, AM-Pharma, Alleviant Medical, Bayer, Beth Israel Deaconess, CardiaWave, CeloNova, Chiesi, Concept Medical, DSI, Duke University, Idorsia Pharmaceuticals, Medtronic, Novartis, Philips; Equity <1% in Applied Therapeutics, Elixir Medical, STEL, CONTROLRAD (spouse); Scientific Advisory Board for AMA, Biosensors (spouse).
Reproduced from: European Heart Journal (2022) 43, 377–386 https://doi.org/10.1093/eurheartj/ehab884, by permission of Oxford University Press on behalf of the European Society of Cardiology
Drug and Material Disclaimer:
The mention of trade names, commercial products organizations, and the inclusion of advertisements in the journal does not imply endorsement by the European Heart Journal, the editors, the editorial board, Oxford University Press or the organization to which the authors are affiliated. The editors and publishers have taken all reasonable precautions to verity drug names and doses, the results of experimental work and clinical findings published in the journal. The ultimate responsibility for the use and dosage of drugs mentioned in the journal and in interpretation of published material lies with the medical practitioner, and the editors and publisher cannot accept liability for damages arising from any error or omissions in the journal. Please inform the editors of any errors.
The opinions expressed in the European Heart Journal are those of the authors and contributors, and do not necessarily reflect those of the European Society of Cardiology, the editors, the editorial board, Oxford University Press or the organization to which the authors are affiliated.
Published on behalf of the European Society of Cardiology. © The Author(s) 2022.
For Permissions, please e-mail: journals.permissions@oup.com
All rights reserved; no part of this publication may be reproduced, stored in a retrieval system, or transmitted in any form or by any means, electronic, mechanical, photocopying, recording, or otherwise without the prior written permission of the Publishers.
Reproduced from: European Heart Journal (2022) 43, 377–386 https://doi.org/10.1093/eurheartj/ehab884
References
1. Piróth Z, Boxma-de Klerk BM, Omerovic E, et al. The natural history of nonculprit lesions in STEMI: an FFR substudy of the compare-acute trial. JACC Cardiovasc Interv 2020; 13: 954–961.
2. Sheth T, Pinilla-Echeverri N, Moreno R, et al. Nonculprit lesion severity and outcome of revascularization in patients with STEMI and multivessel coronary disease. J Am Coll Cardiol 2020; 76: 1277–1286.
3. Puymirat E, Cayla G, Simon T, et al. Multivessel PCI guided by FFR or angiography for myocardial infarction. N Engl J Med 2021; 385: 297–308.
4. Denormandie P, Simon T, Cayla G, et al. Compared outcomes of ST-Elevation myocardial infarction patients with multivessel disease treated with primary percutaneous coronary intervention and preserved fractional flow reserve of non-culprit lesions treated conservatively and of those with low fractional flow reserve managed invasively: insights from the FLOWER MI trial. Circ Cardiovasc Interv 2021; 14: e011314.
5. Cerrato E, Mejía-Rentería H, Dehbi HM, et al. Revascularization deferral of nonculprit stenoses on the basis of fractional flow reserve: 1-year outcomes of 8,579 patients. JACC Cardiovasc Interv 2020; 13: 1894–1903.
6. Montone RA, Niccoli G, Crea F, Jang I-K. Management of non-culprit coronary plaques in patients with acute coronary syndrome. Eur Heart J 2020; 41: 3579–3586.
7. Banning AP, Serruys P, De Maria GL, Ryan N, Walsh S, Gonzalo N, et al. Five-year outcomes after state-of-the-art percutaneous coronary revascularization in patients with de novo three-vessel disease: final results of the SYNTAX II study. Eur Heart J 2021. https://doi.org/10.1093/eurheartj/ehab703
8. Fearon WF, Zimmermann FM, De Bruyne B, et al. Fractional flow reserve–guided PCI as compared with coronary bypass surgery. N Engl J Med 2021. https://doi.org/10.1056/NEJMoa2112299. Published online ahead of print.
9. Takahashi K, Serruys PW, Fuster V, et al. Redevelopment and validation of the SYNTAX score II to individualise decision making between percutaneous and surgical revascularisation in patients with complex coronary artery disease: secondary analysis of the multicentre randomised controlled SYNTAXES trial with external cohort validation. Lancet 2020; 396: 1399–1412.
10. Hara H, Takahashi K, van Klaveren D, et al. Sex differences in all-cause mortality in the decade following complex coronary revascularization. J Am Coll Cardiol 2020; 76: 889–899.
11. Kawashima H, Serruys PW, Ono M, Hara H, O’Leary N, Mack MJ, et al. Impact of optimal medical therapy on 10-year mortality after coronary revascularization. J Am Coll Cardiol 2021; 78: 27–38.
12. Madhavan MV, Howard JP, Naqvi A, et al. Long-term follow-up after ultrathin vs. conventional 2nd-generation drugeluting stents: a systematic review and meta-analysis of randomized controlled trials. Eur Heart J 2021; 42: 2643–2654.
13. Romaguera R, Salinas P, Gomez-Lara J, et al. Amphilimus- versus zotarolimus-eluting stents in patients with diabetes mellitus and coronary artery disease (SUGAR trial). Eur Heart J 2021: hab395.
14. Jeger RV, Farah A, Ohlow M-A, et al. Long-term efficacy and safety of drug-coated balloons versus drug-eluting stents for small coronary artery disease (BASKET-SMALL 2): 3-year follow-up of a randomised, non-inferiority trial. Lancet 2020; 396: 1504–1510.
15. Giacoppo D, Alfonso F, Xu B, et al. Paclitaxel-coated balloon angioplasty vs. drug-eluting stenting for the treatment of coronary instent restenosis: a comprehensive, collaborative, individual patient data meta-analysis of 10 randomized clinical trials (DAEDALUS study). Eur Heart J 2020; 41: 3715–3728.
16. Hildick-Smith D, Egred M, Banning A, et al. The European bifurcation club Left Main Coronary Stent study: a randomized comparison of stepwise provisional vs. systematic dual stenting strategies (EBC MAIN). Eur Heart J 2021; 42: 3829–3839.
17. Jaffer FA, Mehilli J, Escaned J. Left main coronary disease at the bifurcation: should the pendulum swing back towards the provisional stenting approach? Eur Heart J 2021; 42: 3840–3843.
18. Ybarra LF, Rinfret S, Brilakis ES, et al. Definitions and clinical trial design principles for coronary artery chronic total occlusion therapies CTO-arc consensus recommendations. Circulation 2021; 143: 479–500.
19. Wu EB, Brilakis ES, Mashayekhi K, et al. Global chronic total occlusion crossing algorithm: JACC state-of-the-art review. J Am Coll Cardiol 2021; 78: 840–853.
20. Beohar N, Kaltenbach LA, Wojdyla D, et al. Trends in usage and clinical outcomes of coronary atherectomy: a report from the national cardiovascular data registry CathPCI registry. Circ Cardiovasc Interv 2020; 13: e008239.
21. Kinnaird T, Gallagher S, Sharp A, et al. Operator volumes and inhospital outcomes: an analysis of 7,740 rotational atherectomy procedures from the BCIS national database. JACC Cardiovasc Interv 2021; 14: 1423–1430.
22. Hill JM, Kereiakes DJ, Shlofmitz RA, et al. Intravascular lithotripsy for treatment of severely calcified coronary artery disease. J Am Coll Cardiol 2020; 76: 2635–2646.
23. Collison D, Didagelos M, Aetesamur-Rahman M, et al. Poststenting fractional flow reserve vs coronary angiography for optimization of percutaneous coronary intervention (TARGET-FFR). Eur Heart J 2021; 42: 4656–4668.
24. Hong S-J, Mintz GS, Ahn C-M, et al. Effect of intravascular ultrasound–guided drug-eluting stent implantation: 5-year follow-up of the IVUS-XPL randomized trial. JACC Cardiovasc Interv 2020; 13: 62–71.
25. Xu B, Tu S, Song L, et al. Angiographic quantitative flow ratio-guided coronary intervention (FAVOR III China): a multicentre, randomised, sham-controlled trial. Lancet 2021; 398: 2149–2159.
26. Shishikura D, Kataoka Y, Di Giovanni G, et al. Progression of ultrasound plaque attenuation and low echogenicity associates with major adverse cardiovascular events. Eur Heart J 2020; 41: 2965–2973.
27. Stone GW, Maehara A, Ali ZA, et al. Percutaneous coronary intervention for vulnerable coronary atherosclerotic plaque. J Am Coll Cardiol 2020; 76: 2289–2301.
28. Kedhi E, Berta B, Roleder T, et al. Thin-cap fibroatheroma predicts clinical events in diabetic patients with normal fractional flow reserve: the COMBINE OCT–FFR trial. Eur Heart J 2021; 42: 4671–4679.
29. Thiele H, Kurz T, Feistritzer H-J, et al. Comparison of newer generation self-expandable vs. balloon-expandable valves in transcatheter aortic valve implantation: the randomized SOLVE-TAVI trial. Eur Heart J 2020; 41: 1890–1899.
30. Tamburino C, Bleiziffer S, Thiele H, et al. Comparison of self-expanding bioprostheses for transcatheter aortic valve replacement in patients with symptomatic severe aortic stenosis: SCOPE 2 randomized clinical trial. Circulation 2020; 142: 2431–2442.
31. Möllmann H, Holzhey DM, Hilker M, et al. The ACURATE neo2 valve system for transcatheter aortic valve implantation: 30-day and 1-year outcomes. Clin Res Cardiol 2021; 110: 1912–1920.
32. Lansky AJ, Makkar R, Nazif T, et al. A randomized evaluation of the TriGuard™ HDH cerebral embolic protection device to Reduce the Impact of Cerebral Embolic LEsions after TransCatheter Aortiic Valve ImplanTation: the REFLECT I trial. Eur Heart J 2021; 42: 2670–2679.
33. Nazif TM, Moses J, Sharma R, et al. Randomized evaluation of TriGuard 3 cerebral embolic protection after transcatheter aortic valve replacement: REFLECT II. JACC Cardiovasc Interv 2021; 14: 515–527.
34. Leclercq F, Robert P, Akodad M, et al. Prior balloon valvuloplasty versus direct transcatheter aortic valve replacement: results from the DIRECTAVI trial. JACC Cardiovasc Interv 2020; 13: 594–602.
35. Jørgensen TH, Thyregod HGH, Ihlemann N, et al. Eight-year outcomes for patients with aortic valve stenosis at low surgical risk randomized to transcatheter vs. surgical aortic valve replacement. Eur Heart J 2021; 42: 2912–2919.
36. Bleiziffer S, Simonato M, Webb JG, et al. Long-term outcomes after transcatheter aortic valve implantation in failed bioprosthetic valves. Eur Heart J 2020; 41: 2731–2742.
37. Vahanian A, Beyersdorf F, Praz F, et al., 2021 ESC/EACTS Guidelines for the management of valvular heart disease. Eur Heart J 2021: ehab395. https://doi.org/10.1093/eurheartj/ehab395
38. Valgimigli M, Frigoli E, Heg D, et al. Dual antiplatelet therapy after PCI in patients at high bleeding risk. N Engl J Med 2021; 385: 1643–1655.
39. Watanabe. STOPDAPT-2 ACS: one-month dual antiplatelet therapy followed by clopidogrel monotherapy in acute coronary syndrome. Eur Heart J 2021.
40. Giacoppo D, Matsuda Y, Fovino LN, et al. Short dual antiplatelet therapy followed by P2Y12inhibitor monotherapy vs. pro-longed dual antiplatelet therapy after percutaneous coronary intervention with second-generation drug-eluting stents: a systematic review and meta-analysis of randomized clinical trials. Eur Heart J 2021; 42: 308–319.
41. Kirtane AJ, Stoler R, Feldman R, et al. Primary results of the EVOLVE short DAPT study: evaluation of 3-month dual antiplatelet therapy in high bleeding risk patients treated with a bioabsorbable polymer- coated everolimus-eluting stent. Circ Cardiovasc Interv 2021; 14(3): e010144.
42. Mehran R, Cao D, Angiolillo DJ, et al. 3- or 1-month DAPT in patients at high bleeding risk undergoing everolimus-eluting stent implantation. JACC Cardiovasc Interv 2021; 14: 1870–1883.
43. Escaned J, Cao D, Baber U, et al. Ticagrelor monotherapy in patients at high bleeding risk undergoing percutaneous coronary intervention: TWILIGHT-HBR. Eur Heart J 2021; 42: 4624–4634.
44. Kim CJ, Park M-W, Kim MC, et al. Unguided deescalation from ticagrelor to clopidogrel in stabilised patients with acute myocardial infarction undergoing percutaneous coronary intervention (TALOS-AMI): an investigator-initiated, open-label, multicentre, non-inferiority, randomised trial. Lancet 2021; 398: 1305–1316.
45. Koo B-K, Kang J, Park KW, et al. Aspirin versus clopidogrel for chronic maintenance monotherapy after percutaneous coronary intervention (HOST-EXAM): an investigator-initiated, prospective, randomised, open-label, multicentre trial. Lancet 2021; 397: 2487–2496.
46. Montalescot G. Anti-thrombotic strategy to lower all cardiovascular and neurologic ischemic and hemorrhagic events after transaortic valve implantation for aortic stenosis—ATLANTIS. American College of Cardiology Virtual Annual Scientific Session (ACC 2021). 2021.
47. Dangas GD, Tijssen JGP, Wöhrle J, et al. A controlled trial of rivaroxaban after transcatheter aortic-valve replacement. N Engl J Med 2020; 382: 120–129.
48. Nijenhuis VJ, Brouwer J, Delewi R, et al. Anticoagulation with or without clopidogrel after transcatheter aortic-valve implantation. N Engl J Med 2020; 382: 1696–1707.
49. Van Mieghem NM, Unverdorben M, Hengstenberg C, et al. Edoxaban versus vitamin k antagonist for atrial fibrillation after TAVR. N Engl J Med 2021; 385: 2150–2160.
50. Westra J, Andersen BK, Campo G, et al. Diagnostic performance of inprocedure angiography-derived quantitative flow reserve compared to pressure-derived fractional flow reserve: the FAVOR II Europe-Japan study. J Am Heart Assoc 2018; 7: e009603.
51. Garot P, Iriart X, Aminian A, et al. Value of FEops HEARTguide patient-specific computational simulations in the planning of left atrial appendage closure with the Amplatzer Amulet closure device: rationale and design of the PREDICT-LAA study. Open Heart 2020; 7: e001326.